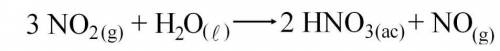Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:20
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
You know the right answer?
2. La siguiente ecuación balanceada representa la segunda etapa del Proceso Ostwald para producir ác...
Questions

Biology, 02.08.2019 23:30

Mathematics, 02.08.2019 23:30

Chemistry, 02.08.2019 23:30

Mathematics, 02.08.2019 23:30


Mathematics, 02.08.2019 23:30


Computers and Technology, 02.08.2019 23:30

Mathematics, 02.08.2019 23:30

Computers and Technology, 02.08.2019 23:30


Computers and Technology, 02.08.2019 23:30

History, 02.08.2019 23:30


History, 02.08.2019 23:30


History, 02.08.2019 23:30