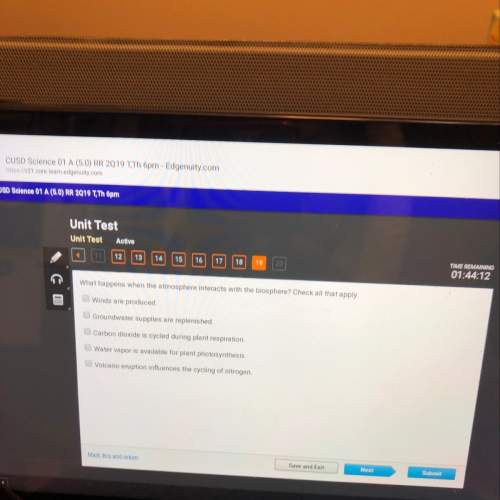
Chemistry, 16.01.2021 04:10 bumpydumper
Please help quick
1) How much heat does 23.0 g of water absorb as its temperature increases from 25.4 °C to 42.8 °C?
2) A sample of unknown metal has a mass of 120.7 g. As the sample cools from 90.5 °C to 25.7 °C, it releases 7020 J of energy. What is the specific heat of the sample?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 22:10
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 23.06.2019 01:40
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1
You know the right answer?
Please help quick
1) How much heat does 23.0 g of water absorb as its temperature increases from 25...
Questions

Biology, 12.08.2020 08:01





Business, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01

Biology, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01



History, 12.08.2020 08:01

History, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01

English, 12.08.2020 08:01




Mathematics, 12.08.2020 08:01

Geography, 12.08.2020 08:01