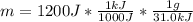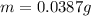Chemistry, 18.01.2021 22:40 Happypuppy1918
A certain chemical reaction releases 31.0 kj/g of heat for each gram of reactant consumed. How can you calculate what mass of reactant will produce of heat

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1
You know the right answer?
A certain chemical reaction releases 31.0 kj/g of heat for each gram of reactant consumed. How can y...
Questions

History, 20.07.2019 22:30

Mathematics, 20.07.2019 22:30


History, 20.07.2019 22:30

Social Studies, 20.07.2019 22:30


Geography, 20.07.2019 22:30

Health, 20.07.2019 22:30

French, 20.07.2019 22:30






Computers and Technology, 20.07.2019 22:30

Mathematics, 20.07.2019 22:30



Mathematics, 20.07.2019 22:30