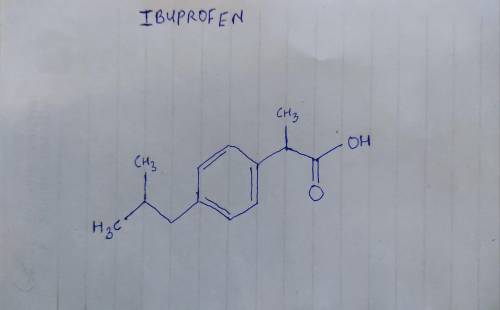
Ibuprofen (aka ADVIL) is a weak acid with a pKa of 4.9. It is absorbed through the stomach and the small intestine as a function of polarity - charged and very polar molecules are absorbed slowly; neutral hydrophobic molecules absorb quickly. If the stomach pH is about 1.5 and the small intestine pH is about 6, where (and why) will more ibuprofen be absorbed into the bloodstream

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 13:30
What would happen if no were added to n(g)+o2=2no(g) at equilibrium?
Answers: 1

Chemistry, 23.06.2019 14:20
Neutral atoms of argon, atomic number 18, have the same number of electrons as each of the following items except: cl- s -2 k+ ca +2 ne
Answers: 2

Chemistry, 23.06.2019 16:00
Which part of the mantle is similar to the crust ? (science earth's layers)
Answers: 1
You know the right answer?
Ibuprofen (aka ADVIL) is a weak acid with a pKa of 4.9. It is absorbed through the stomach and the s...
Questions


Mathematics, 30.03.2021 04:10


Mathematics, 30.03.2021 04:10



Mathematics, 30.03.2021 04:10


Physics, 30.03.2021 04:10


Mathematics, 30.03.2021 04:10



English, 30.03.2021 04:10



Mathematics, 30.03.2021 04:10

History, 30.03.2021 04:10

Mathematics, 30.03.2021 04:10