
Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2


Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2

Chemistry, 23.06.2019 10:00
Why sncl2 is solid while sncl4 is liquid at room temprature explain it in easy way
Answers: 1
You know the right answer?
4.Calculate the volume of 40g of Helium (He) at rtp....
Questions

Mathematics, 19.01.2021 14:00


Physics, 19.01.2021 14:00




Chemistry, 19.01.2021 14:00

Mathematics, 19.01.2021 14:00

Mathematics, 19.01.2021 14:00

Computers and Technology, 19.01.2021 14:00


History, 19.01.2021 14:00

English, 19.01.2021 14:00


Mathematics, 19.01.2021 14:00


Spanish, 19.01.2021 14:00

Mathematics, 19.01.2021 14:00


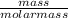
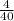 = 0.1mole
= 0.1mole 

