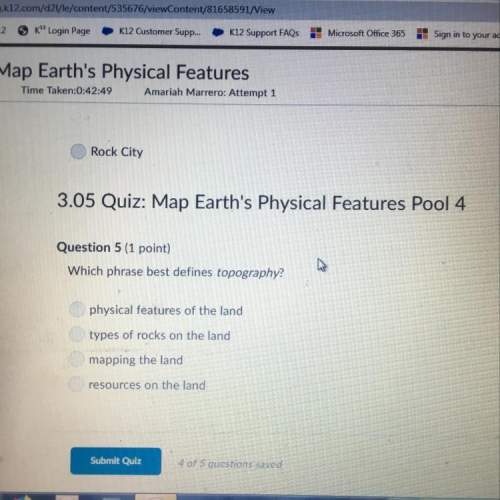
How many electrons are in the outermost energy level of a neutral
Fluorine atom?...

Chemistry, 20.01.2021 05:50 pearljammarow6ujs
How many electrons are in the outermost energy level of a neutral
Fluorine atom?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2
You know the right answer?
Questions

English, 12.04.2020 22:02


Mathematics, 12.04.2020 22:02

Physics, 12.04.2020 22:02

Mathematics, 12.04.2020 22:02




English, 12.04.2020 22:05


English, 12.04.2020 22:16

English, 12.04.2020 22:16


Mathematics, 12.04.2020 22:16

Advanced Placement (AP), 12.04.2020 22:16




Mathematics, 12.04.2020 22:16