
Chemistry, 21.01.2021 22:20 stephensandrew4293
When the concentration of A in the reaction A B was changed from 1.20 M to 0.60 M, the half-life increased from 2.0 min to 4.0 min at 25°C. Calculate the order of the reaction and the rate constant.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1
You know the right answer?
When the concentration of A in the reaction A B was changed from 1.20 M to 0.60 M, the half-life in...
Questions


English, 20.03.2021 01:30




Mathematics, 20.03.2021 01:30

Advanced Placement (AP), 20.03.2021 01:30

History, 20.03.2021 01:30

Physics, 20.03.2021 01:30

Mathematics, 20.03.2021 01:30

Mathematics, 20.03.2021 01:30

Mathematics, 20.03.2021 01:30


Mathematics, 20.03.2021 01:40



History, 20.03.2021 01:40



Biology, 20.03.2021 01:40

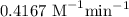
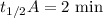
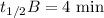
![{[A]_0}_A=1.2\ \text{M}](/tpl/images/1053/6371/ba19d.png)
![{[A]_0}_B=0.6\ \text{M}](/tpl/images/1053/6371/61cc0.png)
![t_{1/2}\propto \dfrac{1}{[A]_0^{n-1}}](/tpl/images/1053/6371/3ebb4.png)
![\dfrac{{t_{1/2}}_A}{{t_{1/2}}_B}=\left(\dfrac{{[A]_0}_B}{{[A]_0}_A}\right)^{n-1}\\\Rightarrow \dfrac{2}{4}=\left(\dfrac{0.6}{1.2}\right)^{n-1}\\\Rightarrow \dfrac{1}{2}=\left(\dfrac{1}{2}\right)^{n-1}](/tpl/images/1053/6371/86904.png)
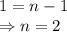
![t_{1/2}=\dfrac{1}{k[A]_0^{n-1}}\\\Rightarrow k=\dfrac{1}{t_{1/2}[A]_0^{n-1}}\\\Rightarrow k=\dfrac{1}{2\times 1.2^{2-1}}\\\Rightarrow k=0.4167\ \text{M}^{-1}\text{min}^{-1}](/tpl/images/1053/6371/f48ea.png)


