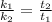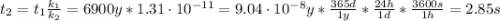Chemistry, 21.01.2021 22:50 DSUDLER5555
The activation energy for a reaction is changed from 184 kJ/mol to 59.0 kJ/mol at 600. K by the introduction of a catalyst. If the uncatalyzed reaction takes about 6900 years to occur, about how long will the catalyzed reaction take

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 07:00
If you used the method of initial rates to obtain the order for no2, predict what reaction rates you would measure in the beginning of the reaction for initial concentrations of 0.200 m, 0.100 m, & 0.050 m no2.
Answers: 3
You know the right answer?
The activation energy for a reaction is changed from 184 kJ/mol to 59.0 kJ/mol at 600. K by the intr...
Questions


History, 31.01.2020 08:48






History, 31.01.2020 08:48

English, 31.01.2020 08:48

Social Studies, 31.01.2020 08:48

English, 31.01.2020 08:48

Mathematics, 31.01.2020 08:48

English, 31.01.2020 08:48

History, 31.01.2020 08:48

World Languages, 31.01.2020 08:48

History, 31.01.2020 08:48

Mathematics, 31.01.2020 08:48



History, 31.01.2020 08:48

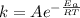
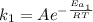 (1)
(1)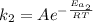 (2)
(2)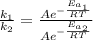
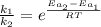
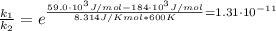
![k = \frac{\Delta [R]}{t}](/tpl/images/1053/9456/cc162.png)
![\frac{k_{1}}{k_{2}} = \frac{\Delta [R]/t_{1}}{\Delta [R]/t_{2}}](/tpl/images/1053/9456/eb896.png)