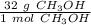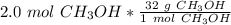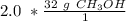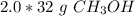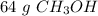Chemistry, 22.01.2021 06:40 dgayles8761
A chemist has 2.0 mol of methanol (CH3OH). The molar mass of methanol is 32.0 g/mol. What is the mass, in grams, of the sample? 16 30 32 64

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3


Chemistry, 23.06.2019 07:50
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1
You know the right answer?
A chemist has 2.0 mol of methanol (CH3OH). The molar mass of methanol is 32.0 g/mol. What is the mas...
Questions

Mathematics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Business, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Physics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

English, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Social Studies, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Health, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

History, 09.09.2020 23:01

Biology, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Mathematics, 09.09.2020 23:01

Social Studies, 09.09.2020 23:01