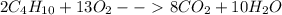Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3


Chemistry, 23.06.2019 07:00
0.88 moles of n2o5 (g) was placed in a sealed 1.00 l vessel. calculate the equilibrium concentration of n2o5. no2, and o2 and the equilibrium constant after equilibrium has been reached by 65.0% of the n2o5 decomposing.
Answers: 1
You know the right answer?
The balanced chemical equation below shows the combustion reaction of butane (C₄H₁₀). What is the mo...
Questions


Mathematics, 05.08.2019 14:10

Mathematics, 05.08.2019 14:10

Mathematics, 05.08.2019 14:10


Mathematics, 05.08.2019 14:10

Mathematics, 05.08.2019 14:10




Biology, 05.08.2019 14:10

Biology, 05.08.2019 14:10

Biology, 05.08.2019 14:10

Mathematics, 05.08.2019 14:10




Mathematics, 05.08.2019 14:10

Computers and Technology, 05.08.2019 15:10

Mathematics, 05.08.2019 15:10