
Chemistry, 25.01.2021 20:20 kameahtravis
A Lewis structure is a two-dimensional representation of a molecule that does not necessarily show what shape that molecule would take in three dimensions. Based on the Lewis structure and your knowledge of VSEPR theory, approximate the smallest bond angle in this molecule.
..
xyx
..

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 23.06.2019 05:30
According to thomson, the atom is a positively charged cloud with electrons scattered throughout. what would the alpha particles do when they hit the foil if thomson were correct
Answers: 1
You know the right answer?
A Lewis structure is a two-dimensional representation of a molecule that does not necessarily show w...
Questions





Mathematics, 18.06.2021 01:20




Mathematics, 18.06.2021 01:20



Mathematics, 18.06.2021 01:30




Mathematics, 18.06.2021 01:30



Mathematics, 18.06.2021 01:30

Computers and Technology, 18.06.2021 01:30

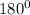
 , the number of electron pairs is 2 that means the hybridization will be
, the number of electron pairs is 2 that means the hybridization will be 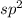 and geometry of the molecule will be linear as there are two bond pairs .The bond angle for linear geometry is 180° which reduces the electron electron repulsions and maximise stability.
and geometry of the molecule will be linear as there are two bond pairs .The bond angle for linear geometry is 180° which reduces the electron electron repulsions and maximise stability.

