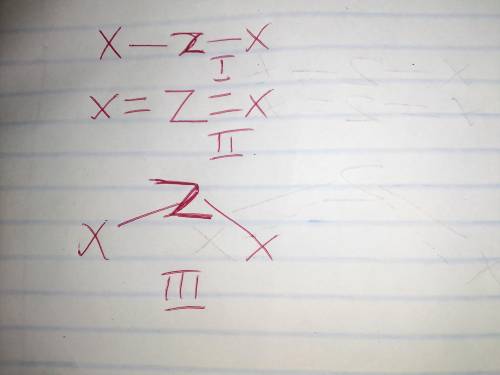Chemistry, 25.01.2021 21:00 juniorlb01
Draw three different Lewis structures that could be possible for ZX2, assuming that Z is the central atom and that X is not hydrogen. Both Z and X obey the octet rule. Give molecular shapes and bond angles for each structure drawn. In each case, use valence shell electron pair repulsion theory to explain why the molecular shape is possible.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2

Chemistry, 23.06.2019 00:10
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3

Chemistry, 23.06.2019 11:00
Which example is a mechanical wave? a.microwave b.radio wave c.water wave d.ultraviolet light
Answers: 1
You know the right answer?
Draw three different Lewis structures that could be possible for ZX2, assuming that Z is the central...
Questions

English, 10.12.2020 18:50



History, 10.12.2020 18:50

Mathematics, 10.12.2020 18:50



Mathematics, 10.12.2020 18:50


Mathematics, 10.12.2020 18:50


Geography, 10.12.2020 18:50


Physics, 10.12.2020 18:50


Mathematics, 10.12.2020 18:50

Chemistry, 10.12.2020 18:50


Mathematics, 10.12.2020 18:50