Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2


Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3
You know the right answer?
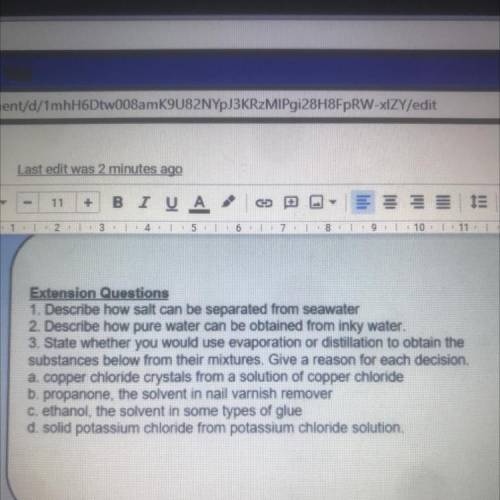
I need help with questions 3a 3b 3c 3d please! In less than 30 minutes
...
...
Questions


Physics, 24.07.2021 17:00


Mathematics, 24.07.2021 17:00

Mathematics, 24.07.2021 17:00



Mathematics, 24.07.2021 17:10



Mathematics, 24.07.2021 17:10

Mathematics, 24.07.2021 17:10

Chemistry, 24.07.2021 17:10


English, 24.07.2021 17:10

Social Studies, 24.07.2021 17:10


Mathematics, 24.07.2021 17:10