Question 10
1 pts
Which of the following statements is true about the mass of an atom?
...

Chemistry, 27.01.2021 20:00 Niktorres560Luckynik
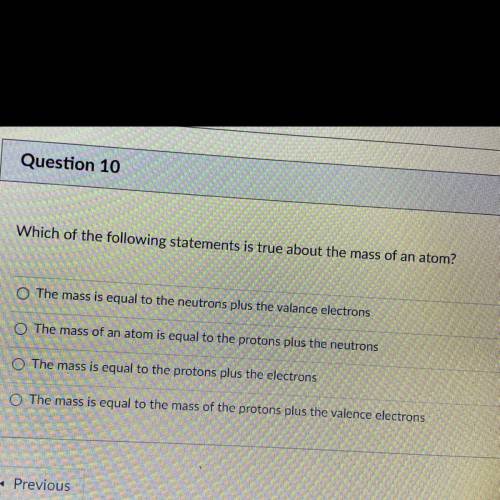
Question 10
1 pts
Which of the following statements is true about the mass of an atom?
The mass is equal to the neutrons plus the valance electrons
The mass of an atom is equal to the protons plus the neutrons
O The mass is equal to the protons plus the electrons
The mass is equal to the mass of the protons plus the valence electrons
+ Previous
Next >

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1


Chemistry, 23.06.2019 13:30
Malik formed a hypothesis that an increase in atmospheric oxygen levels by 10% would cause red-legged grasshoppers to grow larger than normal. suppose that malik performs an experiment to test his hypothesis. which of these actions would represent a scientific mistake in his experiment? a. he experiments on live grasshoppers instead of preserved ones. b. he focuses on red-legged grasshoppers instead of all kinds of grasshoppers. c. he varies the nitrogen and carbon dioxide levels in the air from one trial to the next. d. he conducts the experiment in a controlled lab setting with a lab partner. e. he measures the mass and length of his specimens at the start of each trial.
Answers: 1

Chemistry, 23.06.2019 15:30
Dona wrote the characteristics of two types of galaxies as shown below: type a: has a large flattened core type b: does not have a regular shape which statement is correct? type a is an irregular galaxy and type b is a lens galaxy. type a is a lens galaxy and type b is an irregular galaxy. type a is a spiral galaxy and type b is an elliptical galaxy. type a is an elliptical galaxy and type b is a spiral galaxy.
Answers: 2
You know the right answer?
Questions

Mathematics, 18.07.2019 13:30





Chemistry, 18.07.2019 13:30



Social Studies, 18.07.2019 13:30

Social Studies, 18.07.2019 13:30


History, 18.07.2019 13:30



Computers and Technology, 18.07.2019 13:30



Mathematics, 18.07.2019 13:30


Advanced Placement (AP), 18.07.2019 13:30



