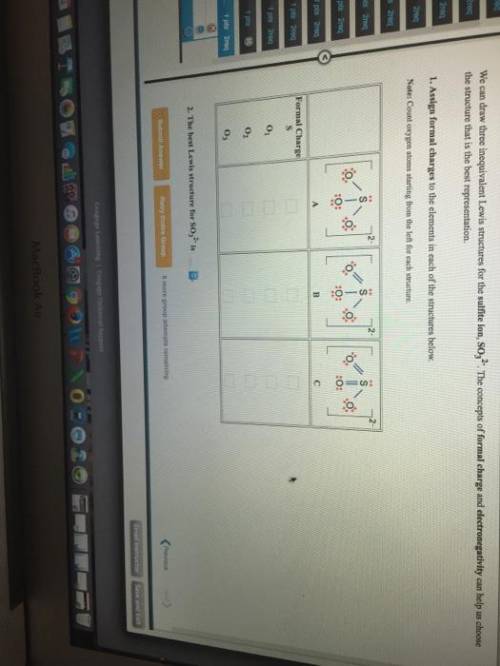
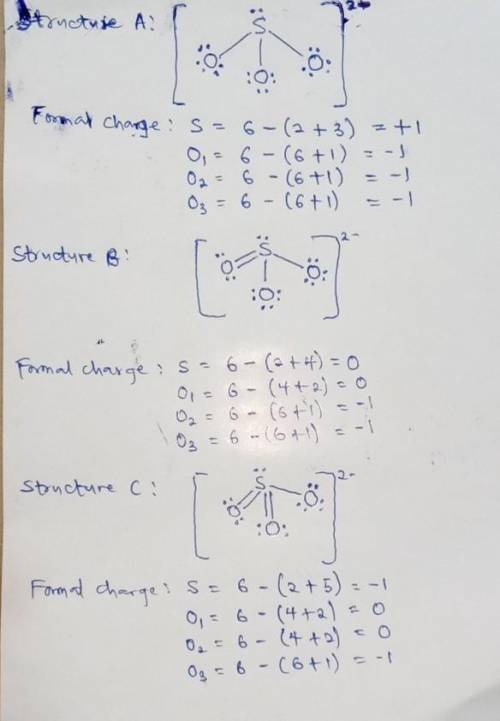
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal charge and electronegativity can help us choose the structure that is the best representation. 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. A B C Formal Charge S O1 O2 O3 2. The best Lewis structure for SO32- is

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 23.06.2019 07:00
How does science use models to gain a better understanding of concepts?
Answers: 1
You know the right answer?
We can draw three inequivalent Lewis structures for the sulfite ion , SO32- . The concepts of formal...
Questions



Mathematics, 31.01.2020 14:59

English, 31.01.2020 14:59

Chemistry, 31.01.2020 14:59



Mathematics, 31.01.2020 14:59

Mathematics, 31.01.2020 14:59

History, 31.01.2020 14:59







Mathematics, 31.01.2020 14:59


Mathematics, 31.01.2020 14:59