Which of the following conclusions can be drawn from the curves in the
energy diagram?
A. The...

Chemistry, 29.01.2021 06:40 hernandezeileen20
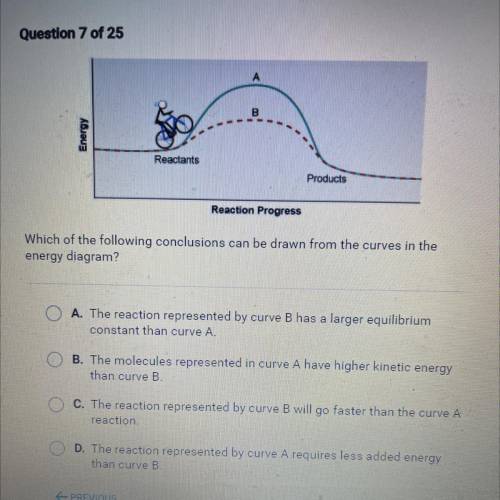
Which of the following conclusions can be drawn from the curves in the
energy diagram?
A. The reaction represented by curve B has a larger equilibrium
constant than curve A.
B. The molecules represented in curve A have higher kinetic energy
than curve B.
C. The reaction represented by curve B will go faster than the curve A
reaction.
D. The reaction represented by curve A requires less added energy
than curve B

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 07:20
Which of the following are acids or bases? 1. sodium hydrogen 2. barium hydroxide solution 3. carbonate solution
Answers: 1

Chemistry, 23.06.2019 08:00
Problem page a jet airplane reaches 846. km/h on a certain flight. what distance does it cover in 13.0 min? set the math up. but don't do any of it. just leave your answer as a math expression. also, be sure your answer includes all the correct unit symbols.
Answers: 2
You know the right answer?
Questions


Computers and Technology, 18.10.2020 08:01



Computers and Technology, 18.10.2020 08:01



English, 18.10.2020 08:01

Mathematics, 18.10.2020 08:01



Mathematics, 18.10.2020 08:01


English, 18.10.2020 08:01



Mathematics, 18.10.2020 08:01



Mathematics, 18.10.2020 08:01



