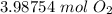CH4 + 202 → CO2 + 2H2O
How many moles of O2 needed to react with 32 grams of CH4?...

Chemistry, 30.01.2021 19:00 winstonbendariovvygn
CH4 + 202 → CO2 + 2H2O
How many moles of O2 needed to react with 32 grams of CH4?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2


You know the right answer?
Questions

Mathematics, 30.01.2022 19:40




Mathematics, 30.01.2022 19:40

Health, 30.01.2022 19:40

Mathematics, 30.01.2022 19:40

Mathematics, 30.01.2022 19:40




Mathematics, 30.01.2022 19:40





Biology, 30.01.2022 19:50


Mathematics, 30.01.2022 19:50

Mathematics, 30.01.2022 19:50

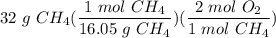 Divide/Multiply:
Divide/Multiply: