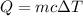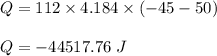What is the total amount of heat released in kilojoules when 112.0 g water at 50.0∘C cools to form ice at −45.0∘C? Use the following values for calculations, as needed.
Properties of Water−−−−−−−−−−−−−−−−−−Specific Heats(∘C)gas=1.84 J/g∘Cliquid=4.184 J/g∘Csolid=2.09 J/g∘C
Heat of VaporizationΔHvap=40.7 kJ/molHeat of FusionΔHfus=6.01 kJ/mol

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
What is the total amount of heat released in kilojoules when 112.0 g water at 50.0∘C cools to form i...
Questions


Mathematics, 16.11.2019 07:31


Mathematics, 16.11.2019 07:31

History, 16.11.2019 07:31

Social Studies, 16.11.2019 07:31

History, 16.11.2019 07:31

Mathematics, 16.11.2019 07:31


History, 16.11.2019 07:31

World Languages, 16.11.2019 07:31



History, 16.11.2019 07:31

French, 16.11.2019 07:31



Spanish, 16.11.2019 07:31


Mathematics, 16.11.2019 07:31