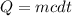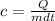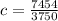Chemistry, 01.02.2021 15:50 brooklynunderwood46
A 250 grams sample of an unknown substance was heated from 35 degree Celsius to 50 degree Celsius. The substance absorbed 7,454 J of energy. What is the specific heat of the substance? Identify if the reaction is exothermic or endothermic

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3

Chemistry, 22.06.2019 06:30
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
A 250 grams sample of an unknown substance was heated from 35 degree Celsius to 50 degree Celsius. T...
Questions

History, 23.06.2020 19:01


Mathematics, 23.06.2020 19:01


Mathematics, 23.06.2020 19:01

English, 23.06.2020 19:01

Mathematics, 23.06.2020 19:01









History, 23.06.2020 19:01

Mathematics, 23.06.2020 19:01