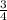Balance this
C3H3N + O2 ⇒ C + H2O + N2...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1


Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1
You know the right answer?
Questions

Biology, 13.10.2019 12:50




Social Studies, 13.10.2019 12:50





Mathematics, 13.10.2019 12:50



English, 13.10.2019 12:50

History, 13.10.2019 12:50

Mathematics, 13.10.2019 12:50


Chemistry, 13.10.2019 12:50

Mathematics, 13.10.2019 12:50


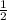 , d =
, d = 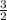 , b =
, b =