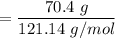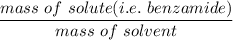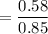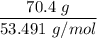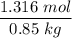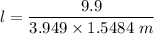Chemistry, 01.02.2021 20:40 alyonaprotopopova
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 2.7 C lower than the freezing point of pure X. On the other hand, when 70.4 g of ammonium chloride (NH CI) are dissolved in the same mass of X, the freezing point of the solution is 9.9 °C lower than the freezing point of pure X.
Required:
Calculate the van't Hoff factor for ammonium chloride in X.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1


Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezin...
Questions

Mathematics, 14.01.2020 22:31


Computers and Technology, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31



Chemistry, 14.01.2020 22:31

Geography, 14.01.2020 22:31


Mathematics, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31

Mathematics, 14.01.2020 22:31

Social Studies, 14.01.2020 22:31



Business, 14.01.2020 22:31



Social Studies, 14.01.2020 22:31