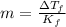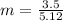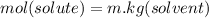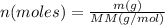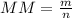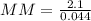Chemistry, 02.02.2021 04:20 Zachary429
When of a certain molecular compound X are dissolved in of benzene , the freezing point of the solution is measured to be . Calculate the molar mass of X. If you need any additional information on benzene, use only what you find in the ALEKS Data resource. Also, be sure your answer has a unit symbol, and is rounded to the correct number of significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2


Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1
You know the right answer?
When of a certain molecular compound X are dissolved in of benzene , the freezing point of the solut...
Questions

Mathematics, 27.01.2020 19:31

Mathematics, 27.01.2020 19:31


Mathematics, 27.01.2020 19:31


Social Studies, 27.01.2020 19:31

Social Studies, 27.01.2020 19:31

History, 27.01.2020 19:31

Mathematics, 27.01.2020 19:31


Mathematics, 27.01.2020 19:31

English, 27.01.2020 19:31


Business, 27.01.2020 19:31


History, 27.01.2020 19:31




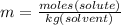
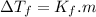
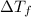 is freezing point depression of solution
is freezing point depression of solution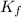 is molal freezing point depression constant
is molal freezing point depression constant