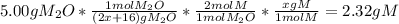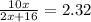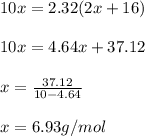Chemistry, 06.02.2021 05:50 chloegrider6713
One mole of a metallic oxide reacts with one mole of hydrogen to produce two moles of the pure metal
and one mole of water. 5.00 g of the metallic oxide produces 2.32 g of the metal. What is the metallic
oxide?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2


Chemistry, 23.06.2019 05:30
Stoichiometry- i need with 14 and 15! an explanation would be appreciated!
Answers: 1
You know the right answer?
One mole of a metallic oxide reacts with one mole of hydrogen to produce two moles of the pure metal...
Questions





Mathematics, 05.02.2021 22:10

History, 05.02.2021 22:10

Arts, 05.02.2021 22:10

Mathematics, 05.02.2021 22:10


Mathematics, 05.02.2021 22:10



Medicine, 05.02.2021 22:10


Computers and Technology, 05.02.2021 22:10



Computers and Technology, 05.02.2021 22:10