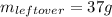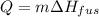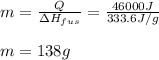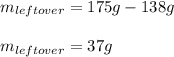Chemistry, 06.02.2021 09:10 gigitorres5803
7. How much ice is left over if 46 kJ of energy is added to 175 g of ice?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 08:30
Of element x has 22 protons, how many electrons does it have
Answers: 1
You know the right answer?
7. How much ice is left over if 46 kJ of energy is added to 175 g of ice?...
Questions



Mathematics, 10.02.2021 04:10



Mathematics, 10.02.2021 04:10

Biology, 10.02.2021 04:10

English, 10.02.2021 04:10

Mathematics, 10.02.2021 04:10

Social Studies, 10.02.2021 04:10



Mathematics, 10.02.2021 04:10



Biology, 10.02.2021 04:10

Mathematics, 10.02.2021 04:10

Computers and Technology, 10.02.2021 04:10

Mathematics, 10.02.2021 04:10