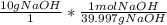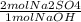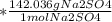Chemistry, 08.02.2021 02:30 ariellake8551
(05.04 MC)
The following reaction shows sodium hydroxide reacting with sulfuric acid.
NaOH + H2SO4 → Na2SO4 + H2O
How many grams of Na2SO4 are produced from 10.0 grams of NaOH?
(Molar mass of Na = 22.989 g/mol, O = 15.999 g/mol, H = 1.008 g/mol, S = 32.065 g/mol) (4 points)
a
17.8 grams
b
19.2 grams
c
35.5 grams
d
38.5 grams
will mark brainliest

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Which statement describes the appearance of a temperature-vs.-time graph? a horizontal line shows that the temperature increases at a constant rate over time. a vertical line shows that the temperature decreases at a constant rate over time. horizontal lines where the temperature is constant during phase changes connect upward-sloping lines where the temperature increases. horizontal lines where the temperature increases are connected by upward-sloping lines where the temperature is constant for each phase.
Answers: 1

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2
You know the right answer?
(05.04 MC)
The following reaction shows sodium hydroxide reacting with sulfuric acid.
N...
N...
Questions


Biology, 01.01.2020 18:31

Biology, 01.01.2020 18:31

Biology, 01.01.2020 18:31


Mathematics, 01.01.2020 18:31


Biology, 01.01.2020 18:31

Mathematics, 01.01.2020 18:31

Mathematics, 01.01.2020 18:31

Health, 01.01.2020 18:31



History, 01.01.2020 18:31




Computers and Technology, 01.01.2020 18:31

Biology, 01.01.2020 18:31