
Chemistry, 08.02.2021 19:10 vondah4014
Sam records the mass of his evaporating dish as 6.488 g. He records the mass of the evaporating dish and the sample of hydrate as 19.72 g. After heating the sample in the evaporating dish to constant weight, the mass of them combined is 11.344 g. How many moles of water were removed from the sample by the heating process?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

Chemistry, 23.06.2019 04:31
Which molecules are more strongly attracted to one another -c3h8o molecules that make up liquid rubbing alcohol or ch4 molecules that make up methane gas
Answers: 3

Chemistry, 23.06.2019 09:20
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of this solution? use the periodic table in the toolbar if needed. 0.0625 g what mass of naoh must be added to the solution to increase the concentration to 30.00% by mass? g
Answers: 2
You know the right answer?
Sam records the mass of his evaporating dish as 6.488 g. He records the mass of the evaporating dish...
Questions

Mathematics, 14.10.2019 15:10






Mathematics, 14.10.2019 15:10



History, 14.10.2019 15:10





Biology, 14.10.2019 15:10

Biology, 14.10.2019 15:10



English, 14.10.2019 15:10

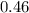 moles of water were removed from the sample by the heating process
moles of water were removed from the sample by the heating process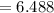 grams
grams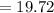 grams
grams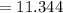 grams
grams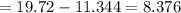 grams
grams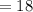 grams
grams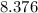 Grams of water is equal to
Grams of water is equal to 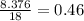 moles
moles

