40 POINTS!
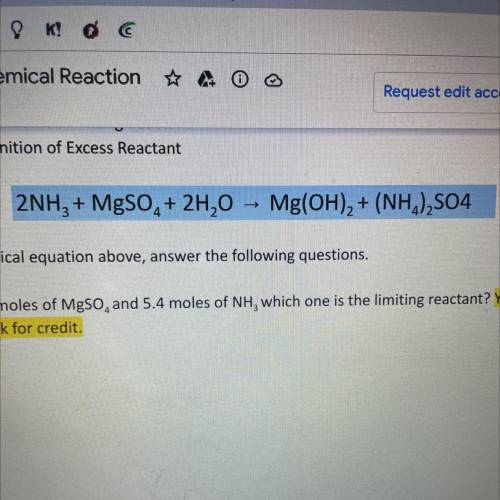
2NH3 + MgSO4 + 2H20 -> Mg(OH)2 + (NH4)2SO4
using the chemical equation ab...

Chemistry, 09.02.2021 01:30 emalvidrez5205
40 POINTS!
2NH3 + MgSO4 + 2H20 -> Mg(OH)2 + (NH4)2SO4
using the chemical equation above answer the following questions. SHOW ALL WORK OR RECEIVE NO CREDIT
A. If I have 4.6 moles of MgSO4 and 5.4 moles of NH3 which one is the limiting reactant? Show work for credit
B. What is the greatest amount of Mg(OH)2 that can be made with 4.6 moles of MgSO4 and 5.4 moles of NH3? Show work for credit
C. How many moles of the excess reactant is left over after the reaction has been completed? Show work for credit

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 10:00
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2
You know the right answer?
Questions

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

History, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Social Studies, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Social Studies, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

History, 14.09.2020 14:01

Spanish, 14.09.2020 14:01

Biology, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01



