
Chemistry, 09.02.2021 01:50 JamesLachoneus
Liquid octane CH3CH26CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 2.3 g of octane is mixed with 12.4 g of oxygen. Calculate the minimum mass of octane that could be left over by the chemical reaction. Round your answer to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2


Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
Liquid octane CH3CH26CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and...
Questions

Health, 19.07.2019 04:30


Mathematics, 19.07.2019 04:30



Social Studies, 19.07.2019 04:30


History, 19.07.2019 04:30

Advanced Placement (AP), 19.07.2019 04:30



Mathematics, 19.07.2019 04:30


Mathematics, 19.07.2019 04:30

Mathematics, 19.07.2019 04:30


Mathematics, 19.07.2019 04:30

Biology, 19.07.2019 04:30

Biology, 19.07.2019 04:30

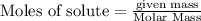
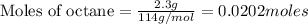
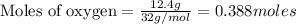
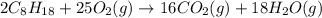
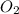
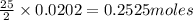 of
of 

