Chemistry, 10.02.2021 17:40 jkenkw4667
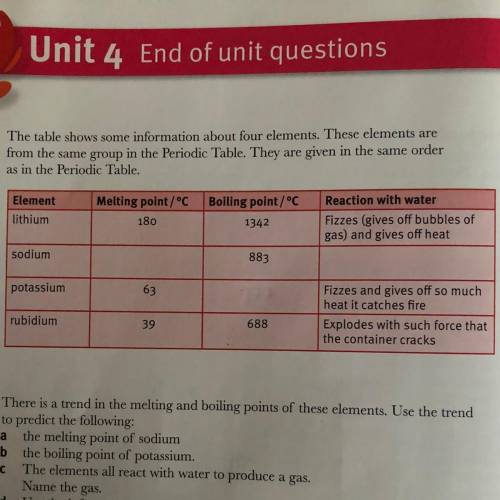
Use the information about the reaction with water to predict the reaction
between sodium and water.
PLEASE HELP ASAP! I will give the brainliest to the correct answer !

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
Use the information about the reaction with water to predict the reaction
between sodium and water....
Questions


Social Studies, 07.07.2019 23:30


Mathematics, 07.07.2019 23:30





Biology, 07.07.2019 23:30

Social Studies, 07.07.2019 23:30

Mathematics, 07.07.2019 23:30

Physics, 07.07.2019 23:30

Mathematics, 07.07.2019 23:30

Physics, 07.07.2019 23:30


Mathematics, 07.07.2019 23:30

Computers and Technology, 07.07.2019 23:30

Mathematics, 07.07.2019 23:30

History, 07.07.2019 23:30