
Chemistry, 16.02.2021 04:40 cooltrey777
The first step in the reaction of Alka–Seltzer with stomach acid consists of one mole of sodium bicarbonate (NaHCO3) reacting with one mole of hydrochloric acid (HCl) to produce one mole of carbonic acid (H2CO3), and one mole of sodium chloride (NaCl). Using this chemical stoichiometry, determine the number of moles of carbonic acid that can be produced from 5 mol NaHCO3 and 9 mol HCl.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1
You know the right answer?
The first step in the reaction of Alka–Seltzer with stomach acid consists of one mole of sodium bica...
Questions



Mathematics, 02.12.2020 22:40



Health, 02.12.2020 22:40

Mathematics, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40



History, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40




History, 02.12.2020 22:40


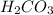 can be produced from 5 mol NaHCO3 and 9 mol HCl.
can be produced from 5 mol NaHCO3 and 9 mol HCl.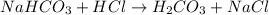
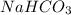 use 1 mole of
use 1 mole of 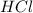
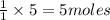 of
of 


