
Chemistry, 18.02.2021 01:00 burritomadness
How do I make the molar masses for 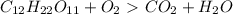 (reactant and products) equal? Please, tell me the steps and how to recalculate.
(reactant and products) equal? Please, tell me the steps and how to recalculate.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

Chemistry, 22.06.2019 17:00
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3

Chemistry, 22.06.2019 19:20
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1
You know the right answer?
How do I make the molar masses for (reactant and products) equal? Please, tell me the steps and how...
Questions

Mathematics, 27.04.2021 20:20


Biology, 27.04.2021 20:20

Computers and Technology, 27.04.2021 20:20






English, 27.04.2021 20:20

Mathematics, 27.04.2021 20:20


Mathematics, 27.04.2021 20:20

Mathematics, 27.04.2021 20:20



Mathematics, 27.04.2021 20:20



History, 27.04.2021 20:20



