1.61 x 104 molecules
Od
2.26 x 1029 molecules
Oe
1.77 x 1029 molecules
Ques...

1.61 x 104 molecules
Od
2.26 x 1029 molecules
Oe
1.77 x 1029 molecules
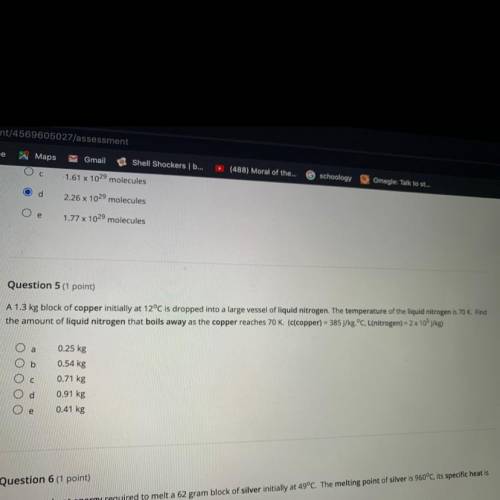
Question 5 (1 point)
A 1.3 kg block of copper initially at 12°C is dropped into a large vessel of liquid nitrogen. The temperature of the liquid nitrogen is 70 K. Find
the amount of liquid nitrogen that boils away as the copper reaches 70 K. (c(copper) = 385 J/kg.°C, L(nitrogen) = 2 x 105 j/kg

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1

Chemistry, 23.06.2019 11:20
Ajar is tightly sealed at 22°c and 772 torr what is the pressure inside a jar after its been heated to 178°c
Answers: 1
You know the right answer?
Questions



English, 04.04.2020 01:30





Mathematics, 04.04.2020 01:30

Health, 04.04.2020 01:30







History, 04.04.2020 01:30

Mathematics, 04.04.2020 01:30

History, 04.04.2020 01:30


English, 04.04.2020 01:30



