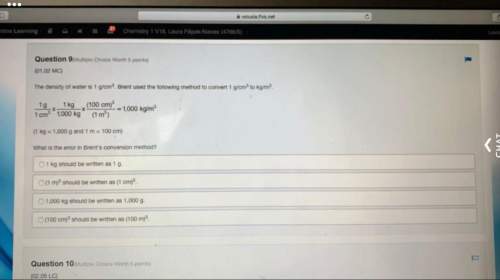
In chemical reactions, a product is
Select one:
any substance that is made from the least num...


Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
Questions




Mathematics, 15.04.2021 04:10

History, 15.04.2021 04:10




Mathematics, 15.04.2021 04:10


History, 15.04.2021 04:10


Mathematics, 15.04.2021 04:10


Mathematics, 15.04.2021 04:10

Mathematics, 15.04.2021 04:10

Biology, 15.04.2021 04:10

Health, 15.04.2021 04:10


Engineering, 15.04.2021 04:10