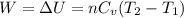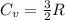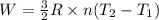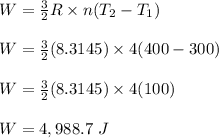Chemistry, 19.02.2021 05:00 helpmewithmath70
4 moles of monoatomic ideal gas is compressed adiabatically causing the temperature to increase from 300 K to 400 K. Calculate the work done on the gas in units of Joules (if the answer is negative, be sure to enter a negative sign in your answer).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
How many molecules of sucrose c12h22o11 are there in 454 grams of sucrose
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 23.06.2019 01:00
You wish to prepare a buffer consisting of acetic acid and sodium acetate with a total acetic acetate plus acetate concentration of 250 mm and a ph of 5. what concentrations of acetic acid and sodium acetate should you use
Answers: 1
You know the right answer?
4 moles of monoatomic ideal gas is compressed adiabatically causing the temperature to increase from...
Questions






Computers and Technology, 13.12.2019 18:31













Computers and Technology, 13.12.2019 18:31