CHEMISTRY SYNTHESIS OF CALCIUM CARBONATE
INTRODUCTION
LABORATORY SIMULATION
1) In this...

Chemistry, 21.02.2021 06:00 lorenaandreahjimenez
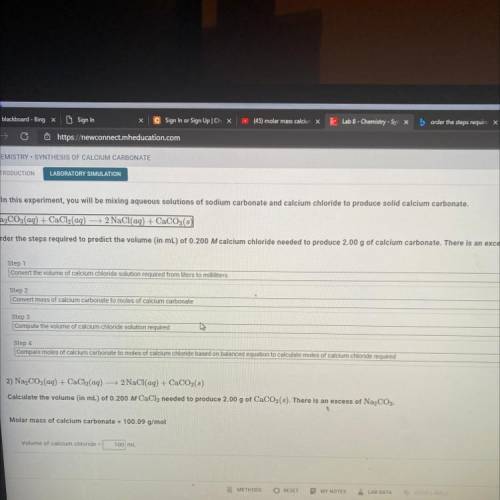
CHEMISTRY SYNTHESIS OF CALCIUM CARBONATE
INTRODUCTION
LABORATORY SIMULATION
1) In this experiment, you will be mixing aqueous solutions of sodium carbonate and calcium chloride to produce solid calcium carbonate.
Na2CO3(aq) + CaCl2(aq) → 2 NaCl(aq) + CaCO3(s)
Order the steps required to predict the volume (in mL) of 0.200 M calcium chloride needed to produce 2.00 g of calcium carbonate. There is al
Step 1
Convert the volume of calcium chloride solution required from liters to milliliters
Step 2
Convert mass of calcium carbonate to moles of calcium carbonate
Step 3
Compute the volume of calcium chloride solution required
Step 4
Compare moles of calcium carbonate to moles of calcium chloride based on balanced equation to calculate moles of calcium chloride required
2) Na2CO3(aq) + CaCl2(aq) 2 NaCl(aq) + CaCO3(8)
Calculate the volume (in mL) of 0.200 M CaCl2 needed to produce 2.00 g of CaCO3(3). There is an excess of Na2CO3.
Molar mass of calcium carbonate = 100.09 g/mol
Volume of calcium chloride =
100 ml
METHODS
RESET
MY NOTES
LAB DATA

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
You know the right answer?
Questions

Social Studies, 22.01.2021 16:30



Mathematics, 22.01.2021 16:40

Mathematics, 22.01.2021 16:40

History, 22.01.2021 16:40




Mathematics, 22.01.2021 16:40




Social Studies, 22.01.2021 16:40


History, 22.01.2021 16:40



English, 22.01.2021 16:40



