Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2


Chemistry, 23.06.2019 01:00
Reactions in cells take place at about a. 40°c b. 0° c. 100°c d. 60°c
Answers: 1
You know the right answer?
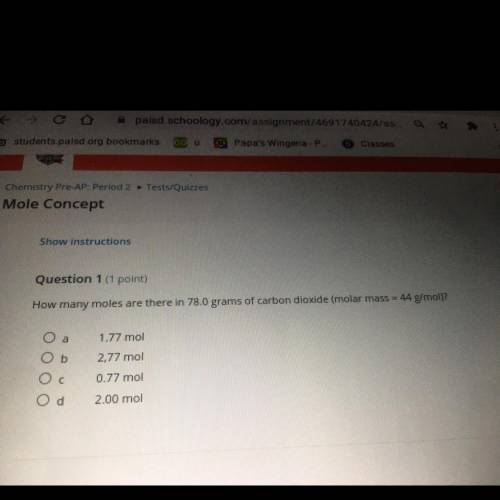
How many moles are there in 3.9 grams of potassium
...
...
Questions





English, 26.03.2021 18:10

Mathematics, 26.03.2021 18:10

Mathematics, 26.03.2021 18:10


Mathematics, 26.03.2021 18:10


Mathematics, 26.03.2021 18:10




History, 26.03.2021 18:10

Law, 26.03.2021 18:10

Health, 26.03.2021 18:10

Mathematics, 26.03.2021 18:10


Mathematics, 26.03.2021 18:10