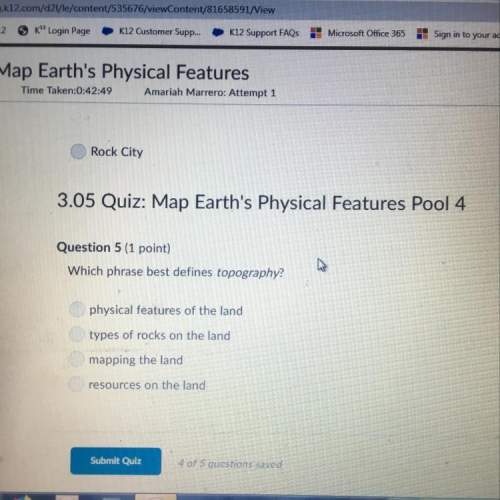
Chemistry, 24.02.2021 22:20 joshuasanders7490
A gas absorbs 56 kJ of heat and does 31 kJ of work. Calculate Δ E.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3


Chemistry, 23.06.2019 06:30
What happens to the glucose molecule during the process of cellular respiration? (5 points) select one: a. it gets broken down. b. it forms oxygen. c. it builds muscles. d. it uses up energy.
Answers: 3
You know the right answer?
A gas absorbs 56 kJ of heat and does 31 kJ of work. Calculate Δ E....
Questions



Health, 30.10.2019 00:31


Mathematics, 30.10.2019 00:31


English, 30.10.2019 00:31

Health, 30.10.2019 00:31





Mathematics, 30.10.2019 00:31

Computers and Technology, 30.10.2019 00:31


Computers and Technology, 30.10.2019 00:31


Mathematics, 30.10.2019 00:31

Mathematics, 30.10.2019 00:31