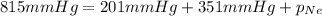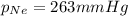A 2.00L mixture of helium, nitrogen, and neon has a total pressure of 815 mmHg at a
temperature of 255K. If the partial pressure of helium is 201 mmHg and the partial
pressure of nitrogen is 351 mmHg, what is the partial pressure of neon in the mixture?
O 709 mmHg
O 512 mmHg
O 667 mmHg
O 263 mmHg

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:50
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x.xx ml
Answers: 1

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2
You know the right answer?
A 2.00L mixture of helium, nitrogen, and neon has a total pressure of 815 mmHg at a
temperature of...
Questions





Mathematics, 07.04.2021 21:40

Mathematics, 07.04.2021 21:40

Mathematics, 07.04.2021 21:40

Social Studies, 07.04.2021 21:40

Mathematics, 07.04.2021 21:40

Mathematics, 07.04.2021 21:40

History, 07.04.2021 21:40

History, 07.04.2021 21:40



Mathematics, 07.04.2021 21:40

Mathematics, 07.04.2021 21:40



Social Studies, 07.04.2021 21:40

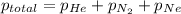
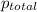 =total pressure of gases = 815 mm Hg
=total pressure of gases = 815 mm Hg
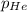 = partial pressure of helium = 201 mm Hg
= partial pressure of helium = 201 mm Hg
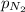 = partial pressure of nitrogen = 351 mm Hg
= partial pressure of nitrogen = 351 mm Hg
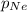 = partial pressure of Neon = ?
= partial pressure of Neon = ?