Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1
You know the right answer?
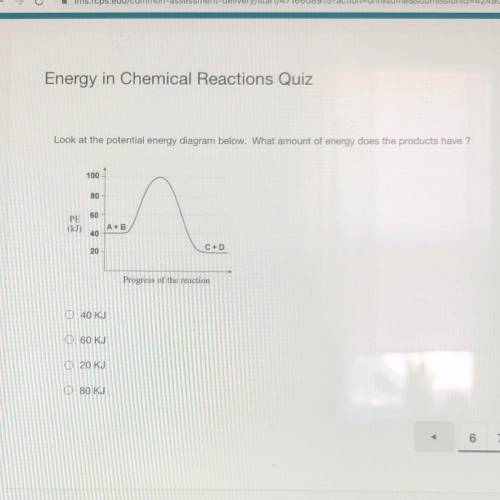
Look at the potential energy diagram below. What amount of energy does the products have ?
100
Questions

Social Studies, 25.11.2021 08:40

Mathematics, 25.11.2021 08:40



Computers and Technology, 25.11.2021 08:40


Computers and Technology, 25.11.2021 08:40







Biology, 25.11.2021 08:40

Advanced Placement (AP), 25.11.2021 08:40



Business, 25.11.2021 08:40

English, 25.11.2021 08:40