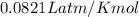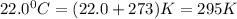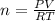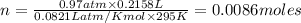Chemistry, 26.02.2021 02:20 jdjdjdjdjjffi7273
2) A student performs this reaction in the laboratory, and collects the hydrogen gas over water. The student collects 215.8 mL of gas. The total pressure is 755.2 mmHg and the temperature is 22.0 oC. c) Use the ideal gas law to calculate how many moles of hydrogen were produced in the reaction

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3

Chemistry, 22.06.2019 15:30
Why does earth rotate? because earth is formed from cold gases collapsing due to gravity because the matter in the nebula that formed earth was spinning because earth forms more than 99% of the mass of the solar system because the hydrogen atoms inside the nebula fused to form helium
Answers: 1

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2
You know the right answer?
2) A student performs this reaction in the laboratory, and collects the hydrogen gas over water. The...
Questions


Health, 31.08.2019 22:50



Mathematics, 31.08.2019 22:50



History, 31.08.2019 22:50



Social Studies, 31.08.2019 22:50



Mathematics, 31.08.2019 22:50


Chemistry, 31.08.2019 22:50


Mathematics, 31.08.2019 22:50


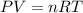
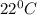 = 19.8 mm Hg
= 19.8 mm Hg