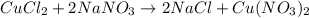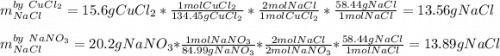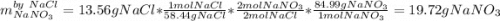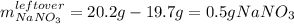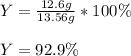Chemistry, 27.02.2021 06:10 imhereforas
If 15.6 grams of copper (ii) chloride react with 20.2 grams of sodium nitrate how many grams of sodium chloride can be formed? What are limiting and excess reactants? How many grams of the excess reactant remain when the reaction stops? If 12.6 grams of sodium chloride are actually produced, what is the percent yield of sodium chloride? (Will mark branliest for best answer)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
9. write the chemical equation for the following word equations. include symbols for physical states in the equation. a. solid zinc sulfide + oxygen gas -> solid zinc oxide + sulfur dioxide gas b. aqueous hydrochloric acid + aqueous barium hydroxide -> aqueous barium chloride + water
Answers: 1


Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3
You know the right answer?
If 15.6 grams of copper (ii) chloride react with 20.2 grams of sodium nitrate how many grams of sodi...
Questions

Mathematics, 10.10.2019 06:00

History, 10.10.2019 06:00

Mathematics, 10.10.2019 06:00

Computers and Technology, 10.10.2019 06:00


English, 10.10.2019 06:00

English, 10.10.2019 06:00



Mathematics, 10.10.2019 06:00


Health, 10.10.2019 06:00

Mathematics, 10.10.2019 06:00



Mathematics, 10.10.2019 06:00

English, 10.10.2019 06:00

Geography, 10.10.2019 06:00

Biology, 10.10.2019 06:00

Social Studies, 10.10.2019 06:00