Choose whether the reaction is exothermic or endothermic.
2C(s) + 2H2(g) → C2H4(9) -Select--
...

Chemistry, 01.03.2021 20:00 godisgoodallthoubxi7
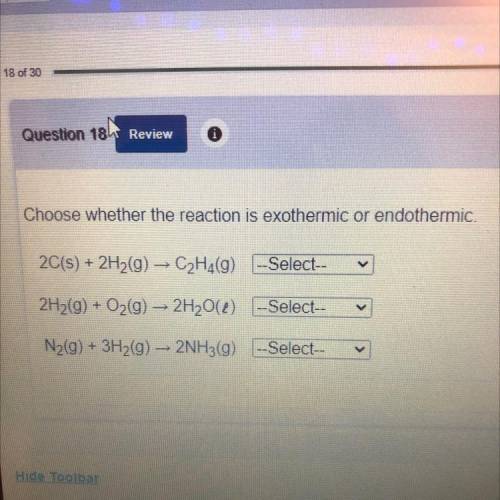
Choose whether the reaction is exothermic or endothermic.
2C(s) + 2H2(g) → C2H4(9) -Select--
2H2(g) + O2(g) → 2H20() -Select--
N2(g) + 3H2(g) + 2NH3(g) --Select--

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2
You know the right answer?
Questions

Business, 12.06.2021 19:00



Mathematics, 12.06.2021 19:00

English, 12.06.2021 19:00


English, 12.06.2021 19:00

History, 12.06.2021 19:00

English, 12.06.2021 19:00

Chemistry, 12.06.2021 19:00

Mathematics, 12.06.2021 19:00

Geography, 12.06.2021 19:00

Mathematics, 12.06.2021 19:00

English, 12.06.2021 19:00

Mathematics, 12.06.2021 19:00

Social Studies, 12.06.2021 19:00

History, 12.06.2021 19:00

Mathematics, 12.06.2021 19:00


Mathematics, 12.06.2021 19:10



