
Chemistry, 02.03.2021 04:10 daigle18383
Sodium-24 has a half-life of 15 hours. What mass in grams of Na-24 will remain in an 18.0 g sample after 90 hours?
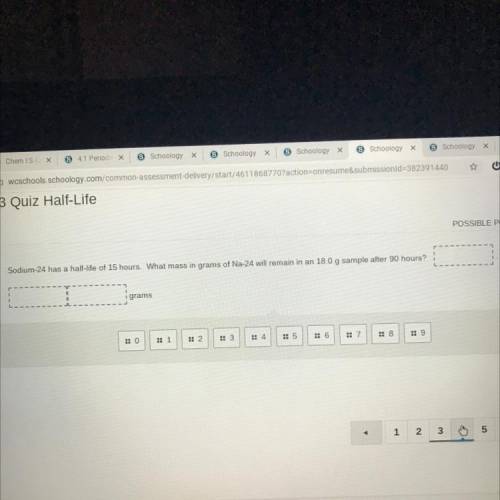

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
Sodium-24 has a half-life of 15 hours. What mass in grams of Na-24 will remain in an 18.0 g sample a...
Questions

History, 21.08.2019 09:00




Mathematics, 21.08.2019 09:00

History, 21.08.2019 09:00


History, 21.08.2019 09:00

Physics, 21.08.2019 09:00

Mathematics, 21.08.2019 09:00

Social Studies, 21.08.2019 09:00


Geography, 21.08.2019 09:00


Chemistry, 21.08.2019 09:00


Health, 21.08.2019 09:00

Mathematics, 21.08.2019 09:00

History, 21.08.2019 09:00

Mathematics, 21.08.2019 09:00



