
Can y’all help me with this last bit of my hw? I don’t really understand it and I would really appreciate it if you could help me.
The questions to the picture is below:
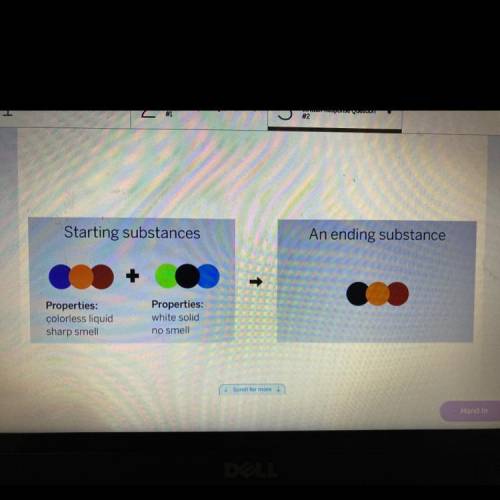
A chemist mixer two substances together: a colorless liquid with a strong strong smell and a white solid with no smell. The substances’ repeating groups of atoms are shown on the left. After they were mixed, the chemist analyzed the results and found two substances, One ending substance had the resorting group of atoms shown above on the right.
Is the ending substance the same as the colorless liquid? What happened to the atoms of the starting substances when the ending substances formed? Be sure to explain your answers to both of these questions.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
Can y’all help me with this last bit of my hw? I don’t really understand it and I would really appre...
Questions



Biology, 08.01.2020 00:31

Social Studies, 08.01.2020 00:31

Computers and Technology, 08.01.2020 00:31





Mathematics, 08.01.2020 00:31





Mathematics, 08.01.2020 00:31







