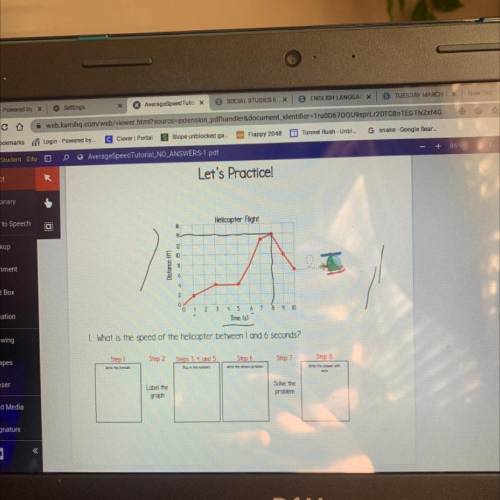
HELPPP DUE AT 11;59 tonight please
...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3
You know the right answer?
Questions


Chemistry, 06.03.2021 07:40


History, 06.03.2021 07:40

Mathematics, 06.03.2021 07:40


Mathematics, 06.03.2021 07:40

World Languages, 06.03.2021 07:40

Mathematics, 06.03.2021 07:40

Mathematics, 06.03.2021 07:40




History, 06.03.2021 07:40

Mathematics, 06.03.2021 07:40


Health, 06.03.2021 07:40

Mathematics, 06.03.2021 07:40


Advanced Placement (AP), 06.03.2021 07:40