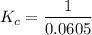Mercury(II) oxide decomposes to form mercury and oxygen, like this:
2Hg (l) + O2 (g) → 2HgO (s)
At a certain temperature, a chemist finds that a reaction vessel containing a mixture of mercury(II) oxide, mercury, and oxygen at equilibrium has the following composition:
compound amount
Hg 14.7g
O2 13.4g
HgO 17.8g
Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits. Clears your work. Undoes your last action. Provides information about entering answers.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2
You know the right answer?
Mercury(II) oxide decomposes to form mercury and oxygen, like this:
2Hg (l) + O2 (g) → 2HgO (s)
Questions


History, 18.04.2020 00:38


History, 18.04.2020 00:38



History, 18.04.2020 00:38

English, 18.04.2020 00:38

Mathematics, 18.04.2020 00:38

Mathematics, 18.04.2020 00:38




Mathematics, 18.04.2020 00:38






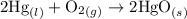
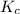 contains aqueous an dgas species only.
contains aqueous an dgas species only. ![$K_c=\frac{1}{[O_2]}$](/tpl/images/1168/7990/eab15.png) ............(1)
............(1)![$[O_2]= \frac{n_{O_2}}{V_{soln}}$](/tpl/images/1168/7990/80b5d.png) ................... (2)
................... (2)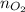 = no. of moles of oxygen gas (mol)
= no. of moles of oxygen gas (mol)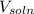 = volume of solution (L)
= volume of solution (L)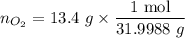
![$[O_2] =\frac{0.418 \ \text{mol}}{6.9 \ \text{L}}$](/tpl/images/1168/7990/d9674.png)