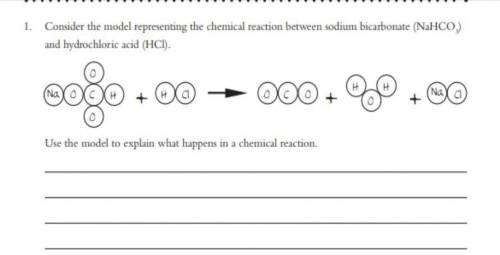
Help?
The question is down below
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 15:00
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1
You know the right answer?
Questions

Mathematics, 03.11.2020 21:20



Advanced Placement (AP), 03.11.2020 21:20




Mathematics, 03.11.2020 21:20

Mathematics, 03.11.2020 21:20



Arts, 03.11.2020 21:20

Chemistry, 03.11.2020 21:20

Mathematics, 03.11.2020 21:20



Mathematics, 03.11.2020 21:20



Spanish, 03.11.2020 21:20