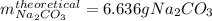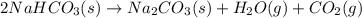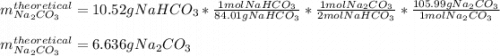Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 04:30
What are the three major branches of natural science? • earth and space science, life science, physical science •earth and space science, physical science, chemistry •physical science, life science, chemistry •life science, chemistry, physics
Answers: 1

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
You know the right answer?
A student heats 10.52 g of sodium hydrogen carbonate in a crucible until the compound completely dec...
Questions


History, 21.09.2019 15:00

English, 21.09.2019 15:00


History, 21.09.2019 15:00

Mathematics, 21.09.2019 15:00

History, 21.09.2019 15:00

Mathematics, 21.09.2019 15:00


World Languages, 21.09.2019 15:00

Mathematics, 21.09.2019 15:00

Social Studies, 21.09.2019 15:00

Spanish, 21.09.2019 15:00




History, 21.09.2019 15:00


Mathematics, 21.09.2019 15:00

Mathematics, 21.09.2019 15:00