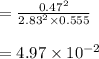Chemistry, 05.03.2021 03:40 eprice32315
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a flask with of sulfur dioxide gas and of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of sulfur trioxide gas to be . Calculate the pressure equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
Questions



Spanish, 08.05.2021 06:00


Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

History, 08.05.2021 06:00



Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

Spanish, 08.05.2021 06:00


Social Studies, 08.05.2021 06:00

Mathematics, 08.05.2021 06:00

 "
"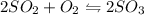

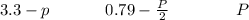
![p= 0.47 \ \ atm\\\\SO_2=3.3-0.47 = 2.83 \ \ atm\\\\O_2= 0.74 -\frac{0.47}{2}=0.74-0.235=0.555 \ atm\\\\K_P=\frac{[PSO_3]^2}{[PSO_2]^2[PO_2]}\\\\](/tpl/images/1170/6349/28d0a.png)