
Chemistry, 05.03.2021 19:00 sydneepollitt32
Carbon dioxide and water react to form methanol and oxygen, like this:
2CO2(g) + 4H2O(g) → 2CH3OH(l) + 3O2 (g)
At a certain temperature, a chemist finds that a 7.5L reaction vessel containing a mixture of carbon dioxide, water, methanol, and oxygen at equilibrium has the following composition:
Compound Amount
CO2 3.28g
H2O 3.86g
CH3OH 1.51g
O2 2.80g
Required:
Calculate the value of the equilibrium constant Kc for this reaction. Round your answer to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:40
Enough of a monoprotic weak acid is dissolved in water to produce a 0.0172 m solution. if the ph of the resulting solution is 2.39 at 20 °c, determine the pka for the acid.
Answers: 1

Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
Carbon dioxide and water react to form methanol and oxygen, like this:
2CO2(g) + 4H2O(g) → 2CH3OH(l...
Questions

Mathematics, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00

History, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00


Mathematics, 10.09.2021 19:00

Computers and Technology, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00

Geography, 10.09.2021 19:00




Mathematics, 10.09.2021 19:00

Mathematics, 10.09.2021 19:00




Mathematics, 10.09.2021 19:00

![\frac{[O_2]^3}{[CO_2]^2[H_2O]^4}](/tpl/images/1172/1518/03065.png)
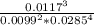 = 24x10³
= 24x10³

